BOSTON – April 18, 2012: Thomas W. Concannon, Ph.D. has developed a framework for assisting investigators in translating their research into medical practice and health policy. His new primer on stakeholder engagement was published this week in the Journal of General Internal Medicine. “A New Taxonomy for Stakeholder Engagement in Patient-Centered Outcomes Research” is a practical how-to for identifying which stakeholders should be involved in the full spectrum of research, and for implementing strategies for their engagement. The paper addresses three questions:
- Who are the stakeholders in PCOR?
- What roles and responsibilities can stakeholders have in PCOR?
- How can researchers start engaging stakeholders?
Tufts investigator Thomas Concannon, PhD, led the project and one of the co-authors is Laurel Leslie, MD, MPH, Director of Tufts CTSI’s Aligning Researchers and Communities for Health (ARCH). The authors are interested in partnering with individuals and organizations to demonstrate and evaluate use of this taxonomy in comparative effectiveness (CER) and patient-centered outcomes research (PCOR). For more information, see the abstract online.
Here is the framework and model presented in the paper:
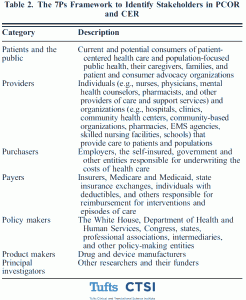
The 7Ps framework identifies key groups to consider for engagement. The first, patients and the public, represents the current and potential consumers of patient-centered health care and population focused public health. The second is providers, including individuals and organizations that provide care to patients and populations. Purchasers, the individuals and entities responsible for underwriting the costs of health care, such as employers, make up the third group. The fourth group consists of payers who are responsible for reimbursement of medical care, such as insurers. The fifth is composed of public policy makers and policy advocates working in the non-governmental sector. Product makers, representing drug and device manufacturers, comprise the sixth group, and principal investigators, or other researchers, make up the seventh.
© Tufts Clinical and Translational Science Institute
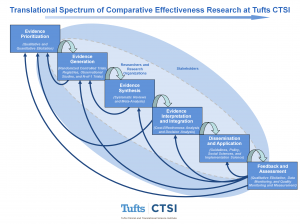
This model illustrates six stages in the translational spectrum of comparative effectiveness research (CER). Each stage is an activity that maybe carried out by researchers and research organizations, as illustrated by the light-shaded oval. Researchers and research organizations are surrounded by stakeholders, as illustrated by the dark-shaded oval. The model illustrates both a sequential flow from evidence prioritization to feedback and assessment as well as a cyclical, iterative process.
